Volume 4 EU Guidelines to GMP Annex 1 44. Outdoor clothing should not be brought into changing rooms leading to grade B and C rooms. For every worker in a grade A/B area, clean sterile (sterilised or adequately sanitised) protective garments should be provided at each work session. Gloves should be regularly disinfected during operations. Masks and gloves should be changed at least for every working session.
How can we monitor this requirement and provide a solution with data integrity ?
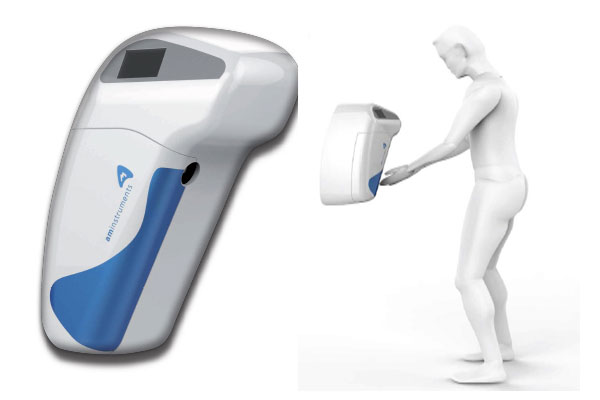
My&Clean and My&Clean+ are contamination-free gloved-hand sanitization devices. The sanitizer is supplied directly from the 1L bottle inside the device. A photocell detects the presence of a gloved hand at a specific distance from the spraying nozzle. My&Clean is specifically developed for sterile cleanroom environments. Once the bottle is placed inside the device, the liquid solution is supplied accordingly, no tubes, no tanks, no cross contamination caused by bottle handling.
My&Clean+ is a system that traces every gloved hand disinfection process including correct execution and frequency by each staff members set programmed requirements. Also, in case of failure, it can signal the necessity of repeating the sanitization. Thanks to this device, it is possible to respect the quality requirement for data integrity in compliance with GMP regulations.My&Clean+ can be installed individually or as multiple interconnected devices, thus recognizing and monitoring specific data related to each singular operator. The liquid is supplied directly from the bottle inside the device, thus avoiding the risk cross contamination and guaranteeing the sterility of the sanitizing product. Also, the quantity of sanitizer available can be inspected on the devices display.
My&Clean and My&Clean+ can be installed as stand-alone device or alternatively can be networked into facility GMP environmental monitoring systems where as to provide data integrity monitoring and door interlock progress post a satisfactory sensitisation cycle.
For more information on this new device, please feel free to email LAF Technologies here